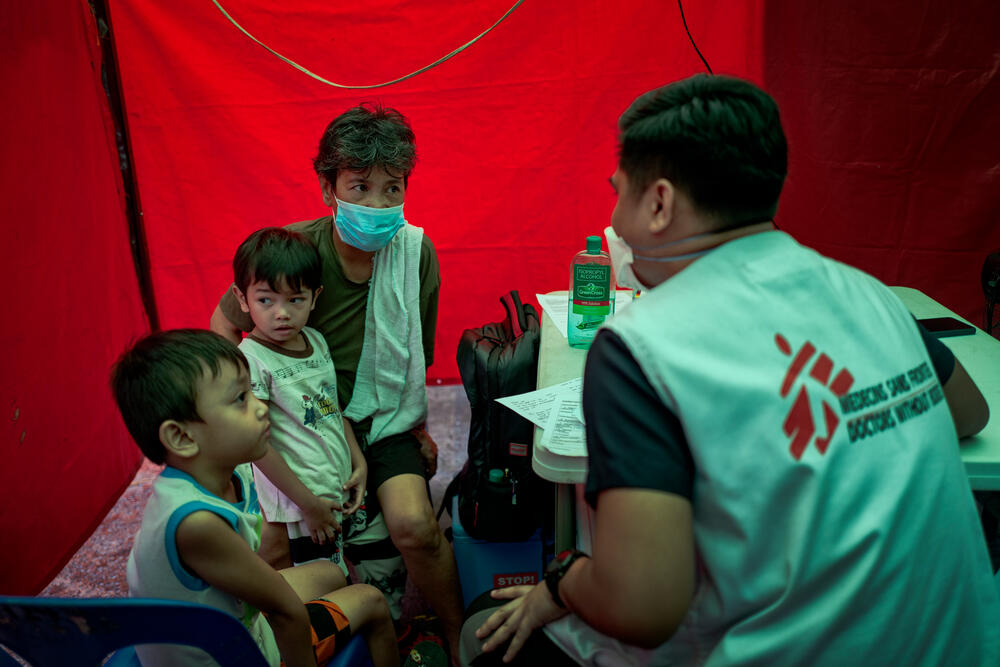
Tuberculosis: MSF calls for urgent access to better testing and treatment
Médecins Sans Frontières / Doctors Without Borders (MSF) is calling on governments and donor organisations to help deliver better care for people with drug-resistant tuberculosis (DR-TB).
This includes speeding up access to new, shorter, safer and more effective treatments*, as well as the diagnostic tests needed to implement the new treatments.
We are also urging the US-based diagnostics corporation Cepheid to drop the price of GeneXpert tests – a tool that is critical to ensuring that people with DR-TB can access this improved care in time.
Ahead of World TB Day, MSF has joined the World Health Organization (WHO) and other organisations to deliver this united 'call to action' for better care.
TB PRACTECAL trial
The WHO issued new guidelines in December 2022 that recommend countries roll out the safer and shorter BPaLM* regimen to treat people with DR-TB, partly based on the results of MSF’s TB PRACTECAL trial.
This was a multi-country, randomised, controlled clinical trial showing that the new, all-oral six-month BPaLM-based treatment is safer and more effective at treating DR-TB than the currently used treatment which is longer, causes intolerable side effects and only cures 60 percent of the people with DR-TB.
“Being able to treat people with drug-resistant TB within six months – instead of up to two years – would be a blessing”
“For me, everything over the past two years has revolved around TB,” says Maria** who underwent two long years of treatment which failed to cure her of DR-TB. She later received the BPaLM regimen in Belarus.
“Living like that was impossible. People [on the old treatment] can’t do anything because they feel awful during the treatment. If only I had started [new] treatment with a short course right away, my life would have been different.”
Critical tests
Access to diagnostic testing for resistance is one of the main hurdles to rolling out the safer and shorter DR-TB treatments.
Currently, the GeneXpert MTB/RIF test made by US corporation Cepheid is the most widely available rapid molecular diagnostic test in countries with high TB rates, able to detect resistance to the ‘first-line’ drug rifampicin.
Despite high sales volumes in affected countries, and MSF’s analysis showing that it costs Cepheid less than $5 (USD) to produce one test, the corporation has kept the price of the test at $9.98 for over a decade now.
Countries should start rolling out the six-month BPaLM regimen to treat DR-TB and ensure nationwide availability of the GeneXpert MTB/RIF tests. If this is not possible, WHO-recommended alternatives such as the Truenat MTB/RIF tests can be used to detect TB and rifampicin resistance so that people with DR-TB can receive this treatment without any delay.
“It is critical that we have better access to tests to diagnose TB and resistance to drugs used for treating TB,” says Stijn Deborggraeve, Diagnostics Advisor at MSF’s Access Campaign.
“[This is] so that we can identify more people who need treatment and roll out the shorter and safer all-oral treatment regimens.
“We yet again call on Cepheid to reduce the price of the TB tests to no more than $5 each, so that more people with drug-resistant TB can be diagnosed in time and be offered improved, lifesaving treatments.”
Drug prices
Drug prices also need to come down further. This could be helped by national TB treatment programmes rolling out these regimens to more people in order to increase demand. This is as well as having more manufacturers supplying affordable generic versions of bedaquiline and pretomanid.
The lowest available price for this newer DR-TB treatment regimen is still $570, and MSF has called for the price of a complete DR-TB course, including the BPaLM regimen, to be no more than $500.
Five countries where MSF works have started implementing the shorter regimens to date, including Belarus, Uzbekistan, Tajikistan, Sierra Leone and Pakistan. Further price reductions will pave the way for the rollout of this treatment in many more countries.
“In a country like Afghanistan, where people are struggling to afford basic food items, travel expenses and medical fees for hospital, being able to treat people with drug-resistant TB within six months – instead of up to two years with the older treatment regimens – would be a blessing,” says Dr Geke Huisman, MSF Medical Coordinator in Afghanistan.
“Access to affordable diagnostic tests remains a major challenge in Afghanistan and other countries in this region because of the high prices of tests.
“Governments, donors and pharmaceutical corporations must act now to ensure an affordable supply of these critical tests and treatments for TB so that more lives can be saved.”
*The six-month BPaLM regimen is composed of bedaquiline (B), pretomanid (Pa), linezolid (L) and moxifloxacin (M). This regimen will not be appropriate for people with TB resistant to bedaquiline, linezolid, pretomanid or delamanid. In parallel with implementing the BPaLM regimen, all countries should urgently scale up access to drug susceptibility testing for the drugs used to treat DR-TB.
** Name changed to protect identity
MSF and tuberculosis
Tuberculosis (TB) is a deadly infectious disease. Every year, over 10 million people develop active TB and 1.5 million die from it.
TB is often thought of as a disease of the past but a recent resurgence and the spread of drug-resistant forms make it very much an issue of the present day and age.